Biotin-tagged probes for MMP expression and activation: design, synthesis, and binding properties
Dragoni, E., Calderone, V., Fragai, M., Jaiswal, R., Luchinat, C., Nativi, C.(2009) Bioconjug Chem 20: 719-727
- PubMed: 19275207
- DOI: https://doi.org/10.1021/bc8003827
- Primary Citation of Related Structures:
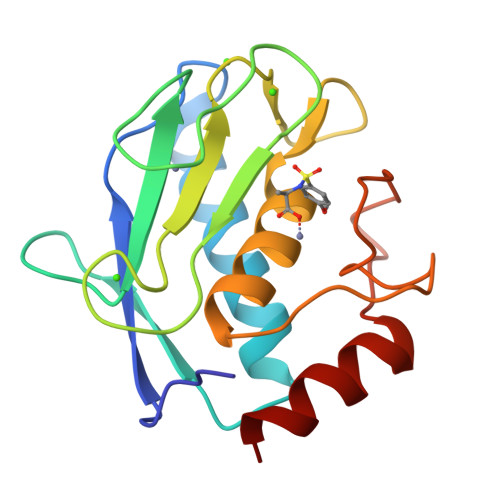
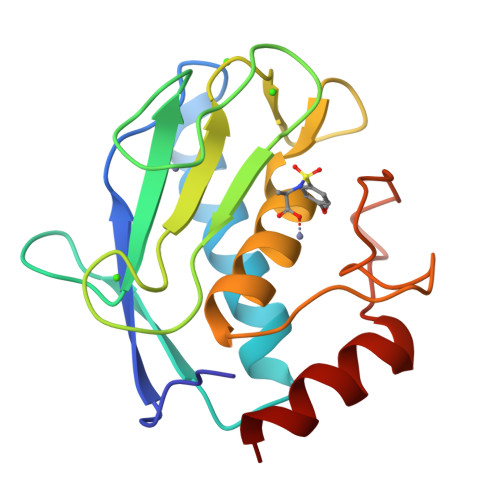
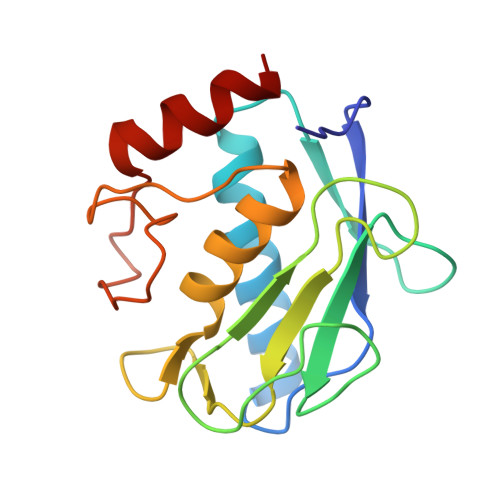
3EHX, 3EHY - PubMed Abstract:
The design and synthesis of biotin chain-terminated inhibitors (BTI) showing high affinity for matrix metalloproteinases (MMPs) on one side and high affinity for avidin through the biotinylated tag on the other are reported. The affinity of the designed BTI toward five different MMPs has been evaluated and the simultaneous formation of a highly stable ternary system Avidin-BTI-MMP clearly assessed. This system will permit the development of new approaches to detect, quantify, or collect MMPs in biological samples, with potential applications in vivo.
Organizational Affiliation:
Magnetic Resonance Center - University of Florence, Sesto Fiorentino, Italy.